Cancers account for nine million of the 18 million deaths each year worldwide. Liver cancer is the 7th most commonly diagnosed cancer and the 4th most common cause of cancer death — over 800,000 deaths annually (lung, breast and bowel cancers being the top three). Viral infection causes about 80% of liver cancers which is why they are more prominent in undeveloped countries. Even so, about 19 people in the UK die every day from liver cancer, the corresponding USA figure being 85, and a gloomy statistic is that the carcinogenic effects of cheap alcohol and unhealthy food have seen a 40% increase in UK liver cancer deaths in the last decade.
Tumours can start in the liver (these are primary cancers that arise, e.g. from viral infection). However, liver metastases are much more common than primary liver cancer — i.e. cancers that have spread from a primary growth elsewhere to form secondary tumours in the liver, i.e. metastasized — bowel cancers being the most frequent source.
Lifting the veil
The topic of metastasis is one of the most intractable and important of all cancer problems — important because it causes most (over 90%) of cancer deaths. In these pages we have followed the slow revelation of how metastatic cells find their way to secondary sites and our current picture owes much to work from David Lyden’s lab in Cornell University, New York. Several blogs have described specific advances (Keeping Cancer Catatonic, Scattering the Bad Seed and Holiday Reading (4) – Can We Make Resistance Futile). These can be summarised as: (1) cells in primary tumours release ‘messengers’ into the circulation that ‘tag’ metastatic sites before any cells actually leave the tumour, (2) the messengers that do the site-tagging are small sacs — extracellular vesicles and particles (EVPs), (3) EVPs find specific addresses by carrying protein labels that home in to different organs — represented in the form of a tube train map in Lethal ZIP Codes, and (4) EVPs help wandering tumour cells to breach the blood-brain barrier (the layer of endothelial cells that encloses the brain), thereby promoting metastasis to the brain (Brainstorming).
Unlimited travel
EVPs released into the circulation from tumours can, in principle, reach any tissue and, following their brain studies, Lyden’s group have now looked at the liver — a major organ involved in many biochemical pathways and, as we have noted, one of the most common cancer sites. With the foregoing picture their results will not surprise. EVPs derived from tumours elsewhere in the body cause massive disruption of liver metabolism. The upshot is a common condition called fatty liver disease. Most people with fatty liver disease have no symptoms but the associated tissue damage can lead to liver cancer (hepatic cancer).
How did they show this?
First they showed in mice that two different types of tumour (melanoma and osteosarcoma), known to preferentially spread to the lung, did not seed liver metastases. However, when they compared the expression levels of different sets of genes in the livers, it was clear that widespread disruption of immune homeostasis (i.e. the balance that maintains immune tolerance) and of metabolism had occurred. Notably the levels of multiple classes of lipids (fatty compounds) were increased in the livers of tumour-bearing mice.
Summary: different types of cancer act from a distance to re-programme liver function via the action of EVPs, ultimately causing cancer. Isolating EVPs and injecting them into tumour-free mice caused similar disruption of liver metabolism, confirming that the effects were due to EVPs and not to tumour cell mestastasis. Major effects were increased inflammatory responses and decreased metabolism (i.e. oxidative phosphorylation and fatty acid metabolism).


Remote primary tumours induce metabolic dysfunction in the liver. Top pair: Liver image from a non-tumour-bearing mouse (left) and (right) liver of a mouse with a tumour (melanoma). Lower pair: Human liver images left: tumour-free individual; right: liver from a patient with pancreatic cancer. Colours: lipid (green) and DNA (blue). From Wang et al. 2023.
A critical finding was that very similar effects were found in the livers from patients with pancreatic ductal adenocarcinoma.
Consequences of dysregulation of liver metabolism
Pursuing these remarkable new findings, Lydens’ group carried out a series of highly detailed studies showing that EVPs cause a particular type of liver cell (Kupffer cells) to secrete tumour necrosis factor — a multifunctional protein (cytokine) that is pro-inflammatory. The microenvironment thus created suppresses fatty acid metabolism and oxidative phosphorylation, thereby promoting fatty liver formation. These livers accumulate huge fat stores that provide a massive energy source for developing cancer cells.
Chemotherapy
Crucially it emerged that dysregulated livers become incapable of metabolizing anti-cancer drugs. For example dacarbazine and doxorubicin, used to treat melanoma and osteosarcoma, respectively, become toxic — in part by reducing the number of red blood cells. Moreover, fatty livers promote cardiovascular disease through increased cholesterol and triglyceride production.
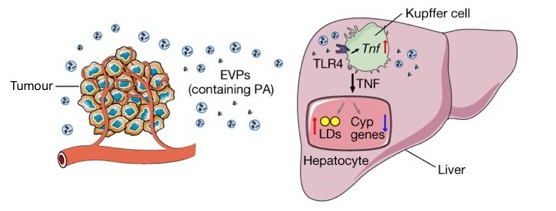
Model for EVP-mediated dysregulation of liver metabolism. Tumour-derived EVPs specifically target Kupffer cells in the liver. EVP-packaged saturated fatty acids, in particular palmitic acid (PA), stimulate the secretion of TNF from Kupffer cells by interacting with TLR4 (toll-like receptor). This produces a pro-inflammatory microenvironment in the liver, thereby inducing fatty liver formation and suppressing the drug-metabolizing activity of the liver (Cyp genes). LDs = lipid droplets. From Wang et al. 2023.
This study is a tour-de-force of applied science. The results are quite breath-taking and open a new aspect on cancer by showing that tumours can alter the function of organs that have not been targeted by metastasis. Perturbing are the facts that this can impact chemotherapy and also link cancers to cardiovascular disease. As ever, however, we can comfort ourselves with the thought that we now know a little more and that this knowledge suggests possible treatment interventions.
Reference
Wang, G., Li, J., Bojmar, L. et al. Tumour extracellular vesicles and particles induce liver metabolic dysfunction. Nature 618, 374–382 (2023). https://doi.org/10.1038/s41586-023-06114-4
